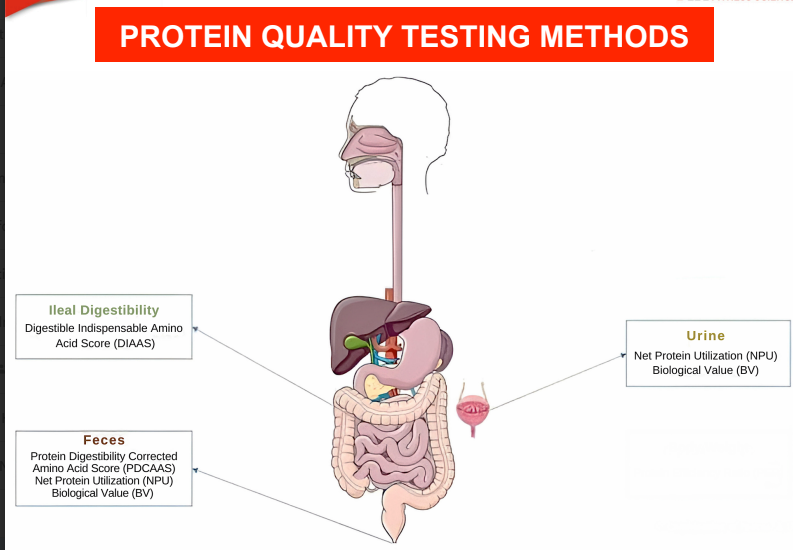
PROTEIN QUALITY TESTING METHODS
1. Biological Value
The concept of Biological Value (BV) was
~100).
2. Protein Efficiency Ratio
The concept of Protein Efficiency Ratio
PDCAAS or DIAAS more reliable measures of protein quality.
3. Net Protein Utilization
The concept of Net Protein Utilization was
for assessing protein quality.
4. Amino Acid Score
The concept of Amino Acid Score (AAS) was
protein quality.
5. Protein Digestibility Corrected Amino Acid Scores:
has limitations, leading to the DIAAS being preferred for accuracy.
6. Digestible Indispensable Amino Acid Scores
The concept of Digestible Indispensable Amino Acid Score (DIAAS) was introduced by
the preferred method by FAO for assessing protein quality.
fecal analysis.
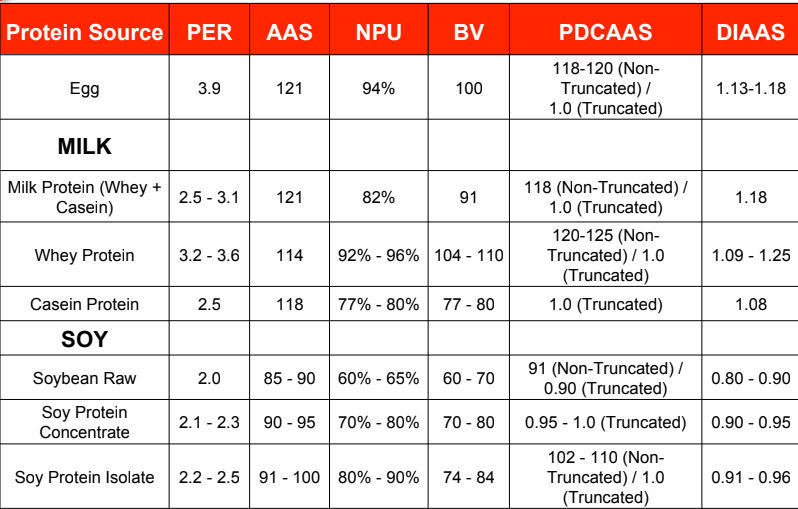
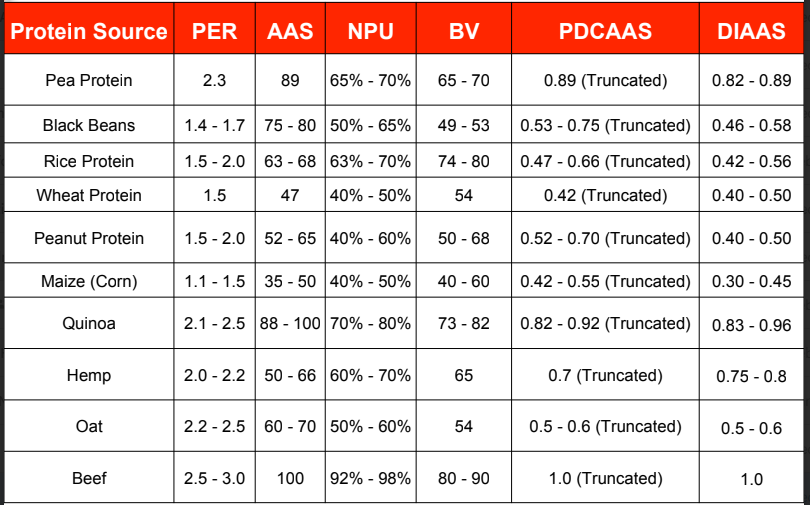
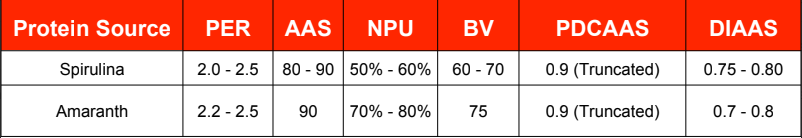
Protein Source

PROTEIN SUPPLEMENT CATEGORIES
1. Standalone Whey Protein Supplement: Whey Protein Concentrate
7. Whey Blends, Plant Protein Blends and Protein Blends


TYPES OF CASEIN SUPPLEMENTS
1. Micellar Casein
: The natural, undenatured form of casein protein
found in milk. Composed of micelles (spherical structures) that form a gel
in the stomach, leading to slow digestion and a gradual/ sustained
release of amino acids. Generally used as a Night-time Protein (before
bed) or In-Between Meals (for satiety).
2. Calcium/ Sodium Caseinate
: Casein that has been treated with
calcium or sodium to form a soluble salt. More processed than Micellar
Casein and is often used in foods, shakes, and supplements because it
mixes well. It is digested faster than Micellar Casein.
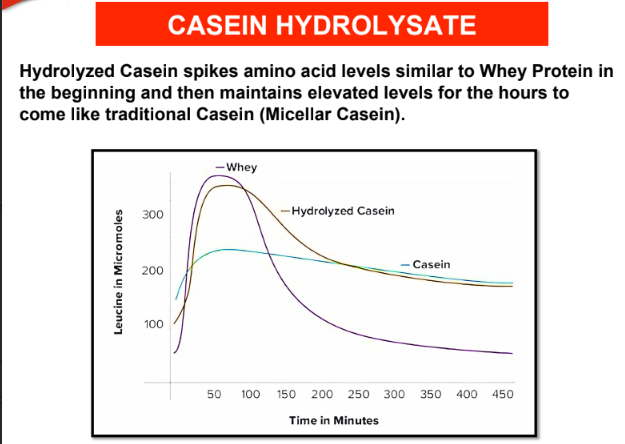
3. Casein Hydrolysate
: Pre-digested casein, also known as casein
hydrolysate, is a form of casein protein that has been broken down into
smaller peptides (chains of amino acids) through a process called
hydrolysis. It is the fastest digesting form of casein.
FILTRATION TECHNIQUES
It is a membrane filtration process used to
It is a membrane filtration process used to
It is a membrane filtration method where
systems like laboratory applications or for initial filtration stages.
It is also known as Tangential-
It is a protein
highly purified whey protein isolates.
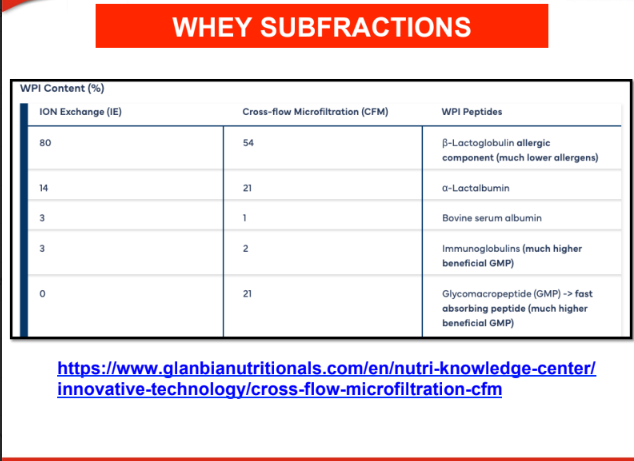
WHEY SUBFRACTIONS
1.Beta-lactoglobulin (β-LG)
2. Alpha-lactalbumin (α-LA)
3. Immunoglobulins (IgG, IgA, IgM):
4. Lactoferrin:
5. Glycomacropeptides (GMP)
• Other minor proteins: Such as serum albumin and enzymes.


Anti-Nutrients in Soy
Trypsin Inhibitors- reduce protein digestibility
Lectins- Can disrupts carbs digestion, binds to the intestinal linin, affecting nutrients
Phytates (Phytic Acid)- Bind to mineral like calcium, iron, zinc reducing their absorption (spinach tomato seeds)
Tannins- Decreases protein digestibility tea
Saponins- Bitter taste, can reduce nutrient availability
Oligosaccharides- causes digestive discomfort (eg, Raffinose, Stachyose)